Recruitment requires consent, a single DNA sample and a very short online CRF
We recruit across all eligible groups and our full eligibility criteria can be found here: Entry criteria
We are approved to take consent in person, by telephone or in the case of retrospective recruitment, consent documentation can also be sent by post.
Our study documents can be found here: Study Documents
For patients lacking capacity - plan a review date to assess for regained capacity.
Regained capacity consent can be followed up by telephone or in writing and we have an ethically approved letter available for this.
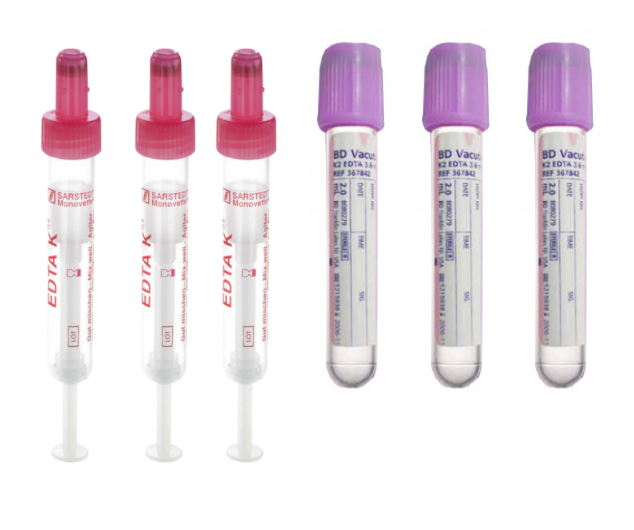
Blood is our preferred method of sampling and can be obtained in any EDTA blood tube. We send 4ml Vacutainer tubes as standard, but can supply monovette tubes if preferred and smaller volume tubes for paediatric and neonatal units.
The amount of blood we need for each sample depends on the patient’s weight:
If a blood sample is not possible saliva may also be obtained from some patients able to use the sample kits independently. Please use only our approved oragene kits for this and ensure enough saliva is collected, right to the fill line.
Dried Blood Spot (DBS) cards are also a sample option available to the paediatric and neonatal units. Contact the GenOMICC team if you are interested in this method of sampling and we’ll supply the DBS cards and instructions.
Take all four of the of GenOMICC barcoded stickers from inside one of the return GenOMICC specimen boxes that we have sent to you.
Label each blood or saliva tube with one of our GCC ID barcoded stickers and put in the plastic bag with absorbent material, seal and package into the GenOMICC specimen box and return to the Wellcome Trust Clinical Reseach Facility (WTCRF), Edinburgh as soon as possible.
The specimen kits are already pre-labelled with a postage paid return address.
Samples will be stable for several days at ambient temperature so there is no problem posting samples on a Friday, over the weekend or during public holidays.
Use one of the patient ID stickers to label the consent form. The patient consent form, REDCap record and sample tube must all be created and labelled with the same GCC Number.
This means we can easily reconcile samples received at the lab, with the correct eCRF.
Destroy any unused stickers - the same number should never be used twice.
Store the consent form in the local site file, which should only be accessible by authorised staff, in a secure area.
RedCap CRF, including: - Clinical and demographic details - Positive microbiology (when available) - 60 day survival outcome
Any EDTA blood tube any size <4ml